Find How Stem Cells Offer New Hope to Arthritis Patients
Stem-cell research is changing how scientists think about arthritis care by targeting inflammation and the joint environment, not just symptoms. This article explains what stem cells are most commonly studied for in arthritis, how clinical trials differ from commercial offerings, what participation typically involves, and how to evaluate trial quality and safety signals. It also covers realistic benefits, limits, and practical questions to ask so readers can explore options with clear expectations.
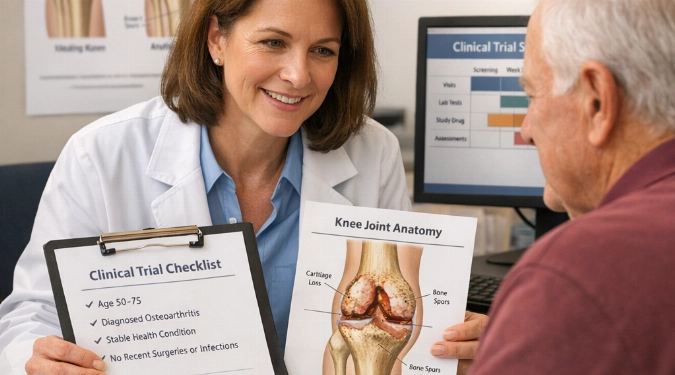
Arthritis can make simple things, like opening a jar or walking to the mailbox, feel like a daily negotiation with pain. Stem-cell research is creating new, carefully tested possibilities that aim to calm inflammation, support tissue repair, and improve function, without promising a cure.
Why Stem Cells Are Being Studied For Arthritis Relief
Arthritis is not one disease. Osteoarthritis is largely driven by cartilage wear and joint degeneration, while rheumatoid arthritis is autoimmune, with the immune system attacking joint tissue. Because stem cells can influence inflammation signals and interact with damaged tissue, researchers are studying whether they can reduce swelling, improve joint mechanics, or slow progression in certain people.
Most arthritis-focused studies involve mesenchymal stromal cells (MSCs), often collected from bone marrow or adipose (fat) tissue, and sometimes from donated birth-tissue sources. MSCs are not “replacement parts” that reliably turn into new cartilage on command; the leading theory is that they release signaling molecules (growth factors, cytokines, extracellular vesicles) that may shift the joint environment toward repair and away from chronic inflammation.
What “Stem Cells For Joint Pain” Usually Means In Research Settings
In many trials, clinicians inject a prepared cell product into a target joint (commonly the knee). Research protocols often track pain scores (such as WOMAC or KOOS), mobility tests, imaging changes on MRI, and safety outcomes over months. Some studies compare cell therapy to placebo injections, hyaluronic acid, platelet-rich plasma (PRP), or physical therapy, which helps clarify what benefit may come from cells specifically.
Timing matters: people with early-to-moderate osteoarthritis may respond differently than those with end-stage “bone-on-bone” damage, where joint replacement is often the standard option. For inflammatory arthritis, the question is different: whether cell-based approaches can safely modulate immune activity without compromising necessary immune defenses.
Where Clinical Trials Fit Versus Commercial Stem-Cell Clinics
Clinical trials are designed to answer specific questions: dosing, preparation method, ideal candidates, how long effects may last, and what adverse events occur. In contrast, many direct-to-consumer clinics offer interventions that may not be well standardized in cell count, viability, or processing. If you are searching phrases like Stem Cell Clinical Trials because you want the most structured oversight, trials typically include defined follow-up schedules, predetermined outcome measures, and formal reporting of side effects.
Safety is a central reason trials matter. Even when a therapy uses your own cells, risks can include infection, bleeding, increased pain or swelling after injection, and rare complications related to the procedure itself. For donated products, additional screening and manufacturing controls may apply, depending on the trial design.
How To Find Legitimate Arthritis Stem-Cell Trials
People often begin with searches such as Stem Cell Clinical Trials Near Me or Arthritis Clinical Trials Nearby Me. A more reliable approach is to verify any study through official registries and academic medical centers, then confirm the details with the study team.
- Check recognized trial registries: ClinicalTrials.gov is the main public registry used by many U.S. studies and lists eligibility criteria, locations, and contacts.
- Look for established research institutions: Universities and hospital systems frequently run osteoarthritis and rheumatoid arthritis trials through orthopedics or rheumatology departments.
- Confirm oversight: Ask whether the trial has Institutional Review Board (IRB) review and what safety monitoring is in place.
- Understand costs and compensation: Trials often cover the investigational product and study visits, but travel, imaging, or labs can vary by protocol.
What Participation Can Look Like
Most studies follow a similar arc: screening, baseline measurements, the treatment (or placebo/comparator) visit, then scheduled follow-ups. Screening may include X-rays or MRI of the joint, lab work, and a medical history review to rule out confounding conditions. Some protocols restrict recent steroid injections, NSAID use, or certain immunosuppressants so results are easier to interpret.
Questions to ask the coordinator include the exact cell source (bone marrow MSCs, adipose-derived MSCs, or allogeneic products), how the product is prepared, how many injections are given, what outcome measures are used (WOMAC, KOOS, VAS pain), and what happens if symptoms worsen during the trial.
Current Reality: Potential Benefits And Clear Limits
Research to date suggests some cell-based approaches may reduce pain and improve function in certain osteoarthritis populations, but results are mixed across studies, and long-term structural cartilage regrowth is not consistently demonstrated. For autoimmune arthritis, evidence is still emerging, and standard disease-modifying treatments remain the backbone of care for many patients.
The most grounded way to view stem-cell research is as a developing option that may complement, not replace, established arthritis management: weight-bearing exercise tailored to tolerance, physical therapy, anti-inflammatory strategies, bracing, injections with known profiles, and surgery when appropriate.
FAQs
Are Stem Cells A Cure For Arthritis?
No. In clinical research, stem-cell interventions are being studied for their ability to reduce symptoms and improve function, and possibly influence inflammation or tissue quality. A cure claim would require consistent, durable disease reversal, which has not been established.
Which Joints Are Most Commonly Studied?
The knee is the most common target in osteoarthritis trials, followed by hip, shoulder, and ankle in smaller studies. Hand arthritis is studied less frequently because of different mechanics and measurement challenges.
How Soon Might Someone Notice Changes In A Trial?
Protocols often measure early changes within weeks, with major checkpoints at about 3, 6, and 12 months. Some participants report gradual improvement over time, while others notice little change, which is why controlled trials are essential.
What Are Common Side Effects Reported?
Commonly tracked issues include temporary post-injection pain, swelling, stiffness, and bruising. Trials also watch for infection, fever, allergic reactions, and unexpected inflammatory flares, even if these are uncommon.
How Can I Tell If A Study Is Well Designed?
Strong signals include clear eligibility criteria, a defined comparator (placebo or standard care), validated outcome measures (like WOMAC or KOOS), transparent follow-up schedules, and a published plan for monitoring adverse events.
Conclusion
Finding how stem cells offer new hope to arthritis patients starts with understanding what the science is truly testing: symptom relief, function, and safety, in carefully monitored settings. If you explore clinical trials, focus on transparent protocols, credible institutions, and clear expectations about what is known and what is still being learned.
Disclaimer: The information provided in this article is for educational and informational purposes only. It does not constitute professional advice. Readers should conduct their own research and consult with qualified professionals before making any decisions.